AG Clauß
AG Clauss - Experimental and translational electrophysiology
In the Clauss group, we investigate mechanisms that lead to arrhythmias. From genetic, molecular and cellular signaling pathways to autonomic and immunological influences, we aim to understand what drives proarrhythmic remodeling. A deeper understanding of these processes will help us to identify the signaling pathways that contribute to the development of arrhythmias and, based on this, to develop and test new therapeutic strategies. The combination of our findings from in vitro, mouse and pig models enables us to translate research results into clinical practice.

Current projects
Myeloid interactions in cardiovascular diseases
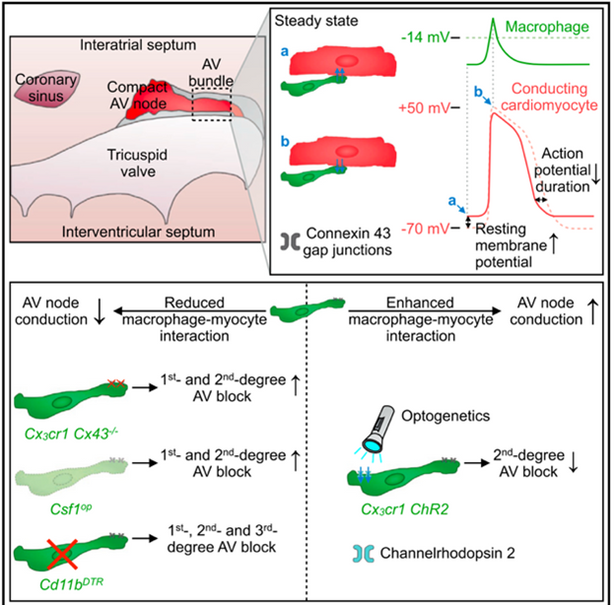
Work of the Clauss group together with our cooperation partners implicates cardiac resident macrophages (CRMs) in physiological conduction through the AV node (Hulsmans & Clauss, Cell, 2017). This results in an electrical coupling of CRMs and cardiomyocytes via connexin-43 gap junctions, whereby CRMs regulate conduction through the AV node.
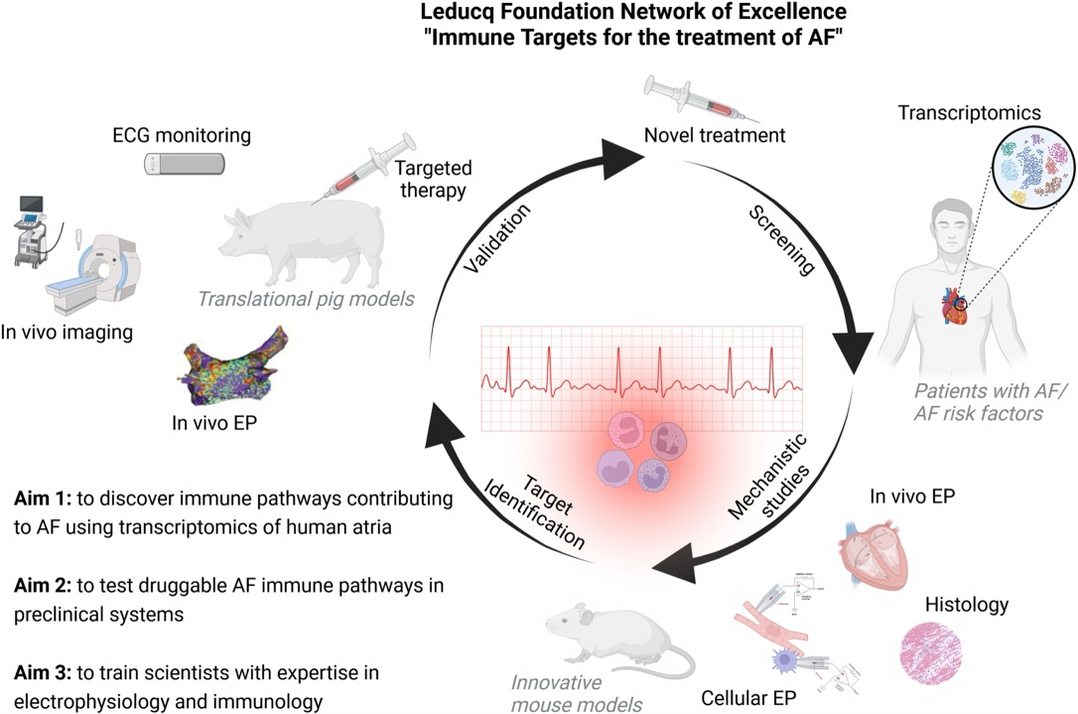
The translation of this discovery to pathological processes identified recruited macrophages as an important factor in the pathogenesis of atrial fibrillation (Hulsmans et al., Science, 2023). However, it remains a major challenge to translate these results from mouse experiments into the clinic. This is partly due to the difference in heart size between mice and humans, which is an important factor in arrhythmia development.
With the help of our HOMER(Hypertension, Obesity, Mitral Regurgitation) pig model for atrial fibrillation, we aim to close the current gaps in the pathogenesis, identify new mechanisms and counteract these with specially developed therapies.
Selected publications
1Clauss S., Nahrendorf M., Kääb S. Leducq-funded program: immune targets for the treatment of atrial fibrillation, Eur. Heart J. 2025 July 1. doi: 10.1093/eurheartj/ehaf176.
2 Xia R, Tomsits P, Loy S, Zhang Z., Pauly V., Schüttler D., Clauss S. Cardiac Macrophages and Their Effects on Arrhythmogenesis. Front Physiol. 2022 June 20. doi:10.3389/fphys.2022.900094
3 Schüttler D., Bapat A., Kääb S., Lee K., Tomsits P., Clauss S.*, Hucker W.* Animal Models of Atrial Fibrillation. Circ Res. 2020 June 18. doi: 10.1161/CIRCRESAHA.120.31636
4Clauss S.*, Schüttler D.*, Bleyer C., Vlcek J., Shakarami M., Tomsits P., Schneider S., Maderspacher F., Chataut K., Trebo A., Wang C., Kleeberger J., Xia R., Baloch E., Hildebrand B., Massberg S., Wakili R., Kääb S. Characterization of a porcine model of atrial arrhythmogenicity in the context of ischaemic heart failure. PLoS One. 2020 May 4. doi: 10.1371/journal.pone.0232374
5Clauss S., Bleyer C., Schüttler D., Tomsits P., Renner S., Klymiuk N., Wakili R., Massberg S., Wolf E., Kääb S. Animal models of arrhythmia: classic electrophysiology to genetically modified large animals. Nat Rev Cardiol. 2019 Mar 20. doi: 10.1038/s41569-019-0179-0.
6 Hulsmans M.*, Clauss S.*, Xiao L.*, Aguirre AD., King KR., Hanley A., Hucker WJ., Wülfers EM., Seemann G., Courties G., Iwamoto Y., Sun Y., Savol AJ., Sager HB., Lavine KJ., Fishbein GA., Capen DE., Da Silva N., Miquerol L., Wakimoto H., Seidman CE., Seidman JG., Sadreyev RI., Naxerova K., Mitchell RN., Brown D., Libby P., Weissleder R., Swirski FK., Kohl P., Vinegoni C., Milan DJ., Ellinor PT., Nahrendorf M. Macrophages Facilitate Electrical Conduction in the Heart. Cell. 2017 April 20. doi: 10.1016/j.cell.2017.03.050
7Clauss S., Wakili R., Hildebrand B., Kääb S., Hoster E., Klier I., Martens E., Hanley A., Hanssen H., Halle M., Nickel T. MicroRNAs as Biomarkers for Acute Atrial Remodeling in Marathon Runners (The miRathon Study - A Sub-Study of the Munich Marathon Study). PLoS One. 2016 Feb 9. doi: 10.1371/journal.pone.0148599
Our methods in the Journal of Visualized Experiments, JoVE
1 Villgrater HE, Xia R, Sharma Chivukula A, Tomsits P, Clauss S. Microdissection and Immunofluorescence Staining of Myocardial Sleeves in Murine Pulmonary Veins. J Vis Exp. 2023(201). doi: 10.3791/65836
2Clauss S. An Innovative Toolkit to Investigate the Complex Mechanisms of Cardiac Arrhythmias. J Vis Exp. 2023(192). doi: 10.3791/64932
3 Tomsits P., Sharma Chivukula A., Raj Chataut K., Simahendra A., Weckbach LT., Brunner S., Clauss S. Real-Time Electrocardiogram Monitoring during Treadmill Training in Mice. J Vis Exp. 2022(183). doi: 10.3791/63873
4 Xia R., Loy S., Kääb S., Titova A., Schulz C., Massberg S., Clauss S. Isolation and Culture of Resident Cardiac Macrophages from the Murine Sinoatrial and Atrioventricular Node. J Vis Exp. 2021(171). doi: 10.3791/62236
5 Tomsits P, Chataut KR, Chivukula AS, Mo L, Xia R, Schüttler D, Clauss S. Analyzing Long-Term Electrocardiography Recordings to Detect Arrhythmias in Mice. J Vis Exp. 2021(171). doi: 10.3791/62386
6 Xia R., Vlcek J., Bauer J., Kääb S., Ishikawa-Ankerhold H., van den Heuvel DA., Schulz C., Massberg S., Clauss S. Whole-Mount Immunofluorescence Staining, Confocal Imaging and 3D Reconstruction of the Sinoatrial and Atrioventricular Node in the Mouse. J Vis Exp. 2020(166). doi: 10.3791/62058
Funded Network
Project: Immune targets for the treatment of atrial fibrillation
2024 - 2029

PD Dr. med. Sebastian Clauß
PI
Jules Hamers (PhD)
Postdoctoral Fellow
Adrian Blair
Postdoctoral Fellow

Dr. med. vet. Julia Vlcek
Veterinarian

Valerie Pauly
Veterinarian and doctoral student (veterinary medicine)
Shasha Zhu
Doctoral student (PhD)
Qifang Wu
Doctoral student (PhD)
Betina Neumann
Doctoral Candidate (PhD)
Xiwen Zhang
Doctoral student (PhD)
Katharina Buchberger
Doctoral student (Dr. med.)
Hannes Villgrater
Doctoral student (Dr. med.)
Stefanie Muschler
Doctoral student (Dr. med.)

Caroline Ballweg
Doctoral student (Dr. med.)
Katherina Klüver
Doctoral student (Dr. med.)

Alina Serter
Doctoral student (Dr. med.)

Theresa Esser
Doctoral student (Dr. med.)
Bianca Hildebrand
Laboratory assistant




