AG Ishikawa-Ankerhold
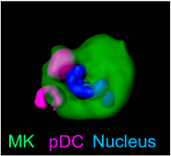
Mechanisms of platelet biogenesis and their impact on immune thrombosis using intravital microscopy
Intravital microscopy(IVM) is the method of choice for the direct observation of dynamic cellular processes in living organisms. The primary goal is to study the interactions between cells in tissues and organs in their natural environment, both under normal physiological conditions and in disease models. This approach provides unique insights that cannot be obtained with any other method at comparable spatial and temporal resolution.
The high-resolution observation of cellular microenvironments in living organisms poses several technological, preparative and methodological challenges. To address these, we employ state-of-the-art 4D in vivo imaging techniques, including confocal laser scanning and multiphoton microscopy, combined with advanced live drift correction software(VivoFollow software 7,12) to correct or minimize motion artifacts caused by respiration and cardiac activity.
In addition to developing the necessary technological and methodological framework, we have also established a number of mouse models that, due to their genetic similarity to humans, facilitate the study of human disease patterns and the development of appropriate therapeutic approaches.
Our research played a central role in the Collaborative Research Center (SFB) 914, which focused on immune cell migration in inflammation, development and disease. There, we provided a core competence platform for 4D multiphoton intravital microscopy of immune cell transport in mouse models. In CRC 1321, which deals with the modeling and control of pancreatic cancer, our work focuses on the investigation of mechanisms that promote coagulation and thrombosis during metastasis. In addition, we participate in numerous collaborative projects, including the SFB 1123, which focuses on atherosclerosis and new therapeutic targets.
An important focus of our research group is the study of immune thrombosis, in particular the mechanisms regulating platelet biogenesis in the bone marrow and their impact on thromboinflammatory diseases. In this context, we have recently discovered an important regulatory mechanism of megakaryopoiesis in which plasmacytoid dendritic cells release interferon-alpha and thus influence platelet production. This groundbreaking discovery, published in Nature 5 (2024), was made possible by the use of IVM and highlights the power of this technology in revealing cell interactions in vivo. Understanding this mechanism provides new insights into the role of immune regulation in platelet formation and its potential contribution to immune thrombotic events.
Our research group is dedicated to the further development and standardization of intravital microscopy techniques as well as the development of highly informative mouse models (link to IVM website). Our scientific focus includes cardiovascular research, immune thrombosis processes and the cellular dynamics and interactions of immune cells with pathogens, cancer cells and within embryonic development.
A major advantage of our work is its close connection to clinical applications. Located at the interface between basic research and clinical medicine, our studies aim to bridge the gap between scientific discovery and medical innovation.
Methods
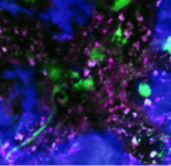
The expertise of AG Ishikawa-Ankerhold includes a wide range of translational mouse models for diseases covering various tissues and organs, including skin, liver, stomach, kidney, spleen, pancreas, placenta, yolk sac, brain, adipose tissue, bone and bone marrow, intestine and muscle tissue(Figure 1).

Figure 1. multiphoton intravital imaging of mouse tissues. Representative intravital multiphoton microscopy (IVM) images of various mouse organs, including brain, bone marrow, liver, stomach, pancreas and yolk sac. The images show immune cells, tumor cells and blood vessels visualized using fluorescent markers. This technique enables real-time tracking of cell dynamics and facilitates the observation of therapeutic interventions in vivo. Different colors represent different structures or cell types and help to investigate tissue-specific interactions and disease progression.
To study these models with high spatial and temporal resolution, we use state-of-the-art intravital imaging techniques, with a focus on 4D multiphoton microscopy and confocal laser scanning microscopy. These technologies allow us to capture dynamic cellular processes in vivo with unprecedented precision.
In addition to advanced imaging, our methodological spectrum includes
Advanced intravital imaging techniques using 4D confocal laser scanning and multiphoton microscopy (link to IVM website)
- 3D printing technologies for customization of sample holders to ensure optimal positioning and stability during imaging
- Image analysis (Imaris, ZEN and Fiji software), multidimensional image processing and image reconstruction to enable a deeper understanding of complex biological structures and interactions
- Immunohistochemical/immunofluorescence characterization of tissues and cell types and use of live cell stains to visualize specific biological processes
- Flow cytometric characterization and separation of different cell populations, enabling targeted analyses and functional studies
- Cell migration and chemotaxis assays using flow chambers, allowing us to study the directional movement of cells in response to different stimuli
By integrating these advanced methods, we aim to refine disease models, improve imaging accuracy and contribute to a deeper understanding of cell behavior in both healthy and diseased individuals.
Publications (selected) *equal contribution
1. Bodogai M, Park B, Braikia FZ, Naqing F, Kumaraswami K, Chen C, Ragonnaud E, Stack S, Ormanns S, Günther M, Ishikawa-Ankerhold H, De S, Ferrucci L, Sen R, Duren Z, Beerman I, Biragyn A. (2025). A distinct population of CD8+ T cells expressing CD39 and CD73 accumulates with age and supports cancer progression. Nat Aging. 2025 Oct;5(10):2055-2069. doi: 10.1038/s43587-025-00966-3
2. Papargyriou A, Najajreh M, Cook DP, Maurer CH, Bärthel S, Messal HA, Ravichandran SK, Richter T, Knolle M, Metzler T, Shastri AR, Öllinger R, Jasper J, Schmidleitner L, Wang S, Schneeweis C, Ishikawa-Ankerhold H, Engleitner T, Mataite L, Semina M, Trabulssi H, Lange S, Ravichandra A, Schuster M, Mueller S, Peschke K, Schäfer A, Dobiasch S, Combs SE, Schmid RM, Bausch AR, Braren R, Heid I, Scheel CH, Schneider G, Zeigerer A, Luecken MD, Steiger K, Kaissis G, van Rheenen J, Theis FJ, Saur D, Rad R, Reichert M. (2025). Heterogeneity-driven phenotypic plasticity and treatment response in branched-organoid models of pancreatic ductal adenocarcinoma. Nat Biomed Eng. 2024 Dec 10. doi: 10.1038/s41551-024-01273-9. Online ahead of print.PMID: 39658630.
3 Blobner J, Dengler L, Eberle C, Herold JJ, Xu T, Beck A, Mühlbauer A, Müller KJ, Teske N, Karschnia P, van den Heuvel D, Schallerer F, Ishikawa-Ankerhold H, Thon N, Tonn JC, Subklewe M, Kobold S, Harter PN, Buchholz VR, von Baumgarten L. (2024). PD-1 blockade does not improve efficacy of EpCAM-directed CAR T-cell in lung cancer brain metastasis. Cancer Immunol Immunother. 2024 Oct 3;73(12):255. doi: 10.1007/s00262-024-03837-9.
4.Ishikawa-Ankerhold HBusch B, Bader A, Maier-Begandt D, Dionisio F, Namineni S, Vladymyrov M, Harrison U, van den Heuvel D, Tomas L, Walzog B, Massberg S, Schulz C, Haas R. (2024). Novel multiphoton intravital imaging enables real-time study of Helicobacter pylori interaction with neutrophils and macrophages in the mouse stomach. PLoS Pathog. 2024 Sep 30;20(9):e1012580. doi: 10.1371/journal.ppat.1012580.
5 Wang X, Campbell B, Bodogai M, McDevitt RA, Patrikeev A, Gusev F, Ragonnaud E, Kumaraswami K, Shirenova S, Vardy K, Alameh MG, Weissman D, Ishikawa-Ankerhold H, Okun E, Rogaev E, Biragyn A. (2024). CD8+ T cells exacerbate AD-like symptoms in mouse model of amyloidosis. Brain Behav Immun. 2024 Nov;122:444-455. doi: 10.1016/j.bbi.2024.08.045. Epub 2024 Aug 25.
6 Gaertner F, Ishikawa-Ankerhold H*, Stutte S, Fu W, Weitz J, Dueck A, Nelakuditi B., Fumagalli V., Van den Heuvel D, Belz L., Sobirova G., Zhang Z, Titova A., Martinez Navarro A., Pekayvaz K., Lorenz M, von Baumgarten L, Jan Kranich, Tobias Straub, Bastian Popper, Vanessa Zheden, Walter Anton Kaufmann, Chenglong Guo, Piontek G., von Stillfried S., Boor P, Colonna M., Clauß S., Schulz C., Brocker T., Walzog B, Scheiermann C., C. Aird W., Nerlov C., Stark K., Petzold T., Engelhardt S., Sixt M., Hauschild R., Rudelius M., Oostendorp R. A. J., Iannacone M., Heinig M., Massberg S. (2024). Plasmacytoid dendritic cells control homeostasis of megakaryopoiesis. NATURE 631:645-653 (2024). https://doi.org/10.1038/s41586-024-07671-y.
7 Mueller T, Pilartz M, Thakur M, LangHeinrich T, Luo J., Block R., Hoeflinger J. K. L, Sarah M., Karaj F., Garcia P. L., Öllinger R., Engleitner T., Thoss J., Voelkl M., Tersteeg C., Koedel U., Zigman K. A., Teupser D., Wygrecka M., Ye H., T Preissner K., Radbruch H., Elezkurtaj S., Mack M., Von Hundelshausen P., Weber C., Massberg S., Schulz C., Rad R., Huber S., Ishikawa-Ankerhold H* and Bernd E*. (2024). Mutual regulation of CD4+ T cells and intravascular fibrin in infections. Haematologica, 2024, 109:2487-2499. doi: https://doi.org/10.3324/haematol.2023.284619.
8 Liu H, Ishikawa-Ankerhold H*, Winterhalter J, Lorenz M, Vladymyrov M, Massberg S, Schulz C, and Orban M. (2023) Multiphoton In Vivo Microscopy of Embryonic Thrombopoiesis Reveals the Generation of Platelets through Budding. Cells 2023, 12(19), 2411; https://doi.org/10.3390/cells12192411
9 Petzold T, Zhang Z, Ballesteros I, Saleh I, Polzin A, Thienel M, Liu L, Ul Ain Q, Ehreiser V, Weber C, Kilani B, Mertsch P, Götschke J, Cremer S, Fu W, Lorenz M, Ishikawa-Ankerhold HRaatz E, El-Nemr S, Görlach A, Marhuenda E, Stark K, Pircher J, Stegner D, Gieger C, Schmidt-Supprian M, Gaertner F, Almendros I, Kelm M, Schulz C, Hidalgo A, Massberg S. (2022) Neutrophil "plucking" on megakaryocytes drives platelet production and boosts cardiovascular disease. Immunity. Oct 18:S1074-7613(22)00542-8. doi: 10.1016/j.immuni.2022.10.001.
10.Ishikawa-Ankerhold H, Kroll J, Heuvel DVD, Renkawitz J, Müller-Taubenberger A. (2022) Centrosome Positioning in Migrating Dictyostelium Cells. Cells. May 29;11(11):1776. https://doi: 10.3390/cells11111776.
11. S. Stutte, H. Ishikawa-Ankerhold, L. Lynch, S. Eickhoff, S. Nasiscionyte, C. Guo, D. van den Heuvel, D. Setzensack, M. Colonna, D. Maier-Begandt, L. Weckbach, T. Brocker, Chr. Schulz, B. Walzog & U. von Andrian. (2022) High-fat diet rapidly modulates the homeostatic phenotype and function of plasmacytoid dendritic cells and alters their trafficking in adipose tissue. J Immunol. 2022 Mar 15;208(6):1445-1455. doi: 10.4049/jimmunol.2100022. epub 2022 Feb 18.
12 Weckbach LT, Schweizer L, Kraechan A, Bieber S, Ishikawa-Ankerhold HHausleiter J, Massberg S, Straub T, Klingel K, Grabmaier U, Zwiebel M, Mann M, Schulz C; EMB Study Group. (2022) Association of Complement and MAPK Activation With SARS-CoV-2 Associated Myocardial Inflammation. AMA Cardiol. 2022 Mar 1;7(3):286-297. doi: 10.1001/jamacardio.2021.5133.
13 Lasch M, Vladymyrov M, van den Heuvel D, Götz P, Deindl E, Ishikawa-Ankerhold H. (2021) Multiphoton intravital imaging as a tool for monitoring leukocyte recruitment during arteriogenesis in vivo in a murine hindlimb model. J Vis Exp. 2021 Sep 30:(175). DOI: 10.3791/62969.
14. Zhang W, Karschnia P, von Mücke-Heim IA, Mulazzani M, Zhou X, Blobner J, Mueller N, Teske N, Dede S, Xu T, Thon N, Ishikawa-Ankerhold HStraube A, Tonn JC, von Baumgarten L. (2021) In vivo two-photon characterization of tumor-associated macrophages and microglia (TAM/M) and CX3CR1 during different steps of brain metastasis formation from lung cancer. Neoplasia. 2021 Nov;23(11):1089-1100.
15 Nicolai L, Kaiser R, Escaig R, Hoffknecht ML, Anjum A, Leunig A, Pircher J, Ehrlich A, Lorenz M, Ishikawa-Ankerhold H, Aird WC, Massberg S, Gaertner F. (2021) Single platelet and megakaryocyte morpho-dynamics uncovered by multicolor reporter mouse strains in vitro and in vivo. Haematologica. 2021 Sep 16.
16 Stutte S, Ruf J, Kugler I, Ishikawa-Ankerhold HParzefall A, Marconi P, Maeda T, Kaisho T, Krug A, Popper B, Lauterbach H, Colonna M, von Andrian U, Brocker T. (2021) Type I interferon mediated induction of somatostatin leads to suppression of ghrelin and appetite thereby promoting viral immunity in mice. Brain Behav Immun. Jul; 95:429-443.
17 Lasch M, Kumaraswami K, Nasiscionyte S, Kircher S, van den Heuvel D, Meister S, Ishikawa-Ankerhold H*, Deindl E*. (2020) RNase A Treatment Interferes With Leukocyte Recruitment, Neutrophil Extracellular Trap Formation, and Angiogenesis in Ischemic Muscle Tissue. Front Physiol. Nov 6, 11: 576736.
18 Weinberger T, Esfandyari D, Messerer D, Percin G, Schleifer C, Thaler R, Liu L, Stremmel C, Schneider V, Vagnozzi RJ, Schwanenkamp J, Fischer M, Busch K, Klapproth K, Ishikawa-Ankerhold HKlösges L, Titova A, Molkentin JD, Kobayashi Y, Engelhardt S, Massberg S, Waskow C, Perdiguero EG, Schulz C. (2020) Ontogeny of arterial macrophages defines their functions in homeostasis and inflammation. Nat Commun. Sept 11, 11: 4549.
19 Stocker TJ, Pircher J, Skenderi A, Ehrlich A, Eberle C, Megens RTA, Petzold T, Zhang Z, Walzog B, Müller-Taubenberger A, Weber C, Massberg S, Ishikawa-Ankerhold H*, Schulz* C. (2018). The Actin Regulator Coronin-1A Modulates Platelet Shape Change and Consolidates Arterial Thrombosis. Thromb Haemost. Dec; 118(12):2098-2111.
20 Pircher J, Czermak T, Ehrlich A, Eberle C, Gaitzsch E, Margraf A, Grommes J, Saha P, Titova A, Ishikawa-Ankerhold HStark K, Petzold T, Stocker T, Weckbach LT, Novotny J, Sperandio M, Nieswandt B, Smith A, Mannell H, Walzog B, Horst D, Soehnlein O, Massberg S, Schulz C. (2018). Cathelicidins prime platelets to mediate arterial thrombosis and tissue inflammation. Nat Commun. Apr 18; 9 (1): 1523.
21 Stremmel C, Schuchert R, Wagner F, Thaler R, Weinberger T, Pick R, Mass E, Ishikawa-Ankerhold HMargraf A, Hutter S, Vagnozzi R, Klapproth S, Frampton J, Yona S, Scheiermann C, Molkentin JD, Jeschke U, Moser M, Sperandio M, Massberg S, Geissmann F, Schulz C. (2018). Yolk sac macrophage progenitors traffic to the embryo during defined stages of development. Nat Commun. Jan 8; 9 (1): 75.
22 Gaertner F, Ahmad Z, Rosenberger G, Fan S, Nicolai L, Busch B, Yavuz G, Luckner M, Ishikawa-Ankerhold HHennel R, Benechet A, Lorenz M, Chandraratne S, Schubert I, Helmer S, Striednig B, Stark K, Janko M, Böttcher RT, Verschoor A, Leon C, Gachet C, Gudermann T, Mederos Y Schnitzler M, Pincus Z, Iannacone M, Haas R, Wanner G, Lauber K, Sixt M, Massberg S. (2017). Migrating platelets are mechano-scavengers that collect and bundle bacteria. Cell. Nov 30; 171 (6): 1368-1382.e23.
23 Stocker TJ, Ishikawa-Ankerhold H, Massberg S, Schulz C (2017). Small but mighty: Platelets as central effectors of host defense. Thromb Haemost. Apr 3;117(4):651-661. doi: 10.1160/TH16-12-0921. Epub 2017 Feb 16
24 Schulz C, Petzold T, Ishikawa-Ankerhold H. (2021) Macrophage regulation of granulopoiesis and neutrophil functions. Antioxid Redox Signal. Oct 27. doi: 10.1089/ars.2020.8203. PMID: 33107319. Review
Book chapters
Grants
Promotion of Women Scientists
Project: Mechanisms regulating splenic platelets biogenesis in sepsis and its implications for cardiovascular diseases
2025 -2027
FöFoLe (funding program for research and teaching of the Medical Dean's Office)-PhD project
Project: Investigating the Role of Plasmacytoid Dendritic Cells in Immune-Primed Platelet Formation in Thromboinflammation
2026 -2027

PD Dr. rer. nat. Hellen Ishikawa-Ankerhold
PI and Head of Multiphoton Intravital Microscopy
Larissa Belz, M. Sc.
PhD student
Gulnoza Sobirova
MD student
Maximilian Seeberger
MD
Yinfeng Zhu
PhD student
Qiongjie Ding
PhD student
Jinlong Wan
PhD student
Paulina Luna David
Med student

Dominic van den Heuvel
technical assistant - light microscopy and image analysis
Sanjana Balaji
Student assistant



